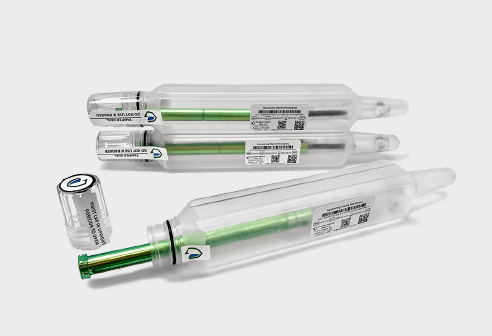

After investing significant time and money into developing a medical device, the importance of its packaging cannot be overlooked. The FDA considers both the device and its packaging as part of the final product, meaning packaging design is critical to the overall device performance. While pouches and trays are commonly used for medical device packaging, tube packaging may be the alternative you’ve been looking for. For this post, we spoke with Guardian Medical, a trusted leader in sterile barrier tube packaging, to share their insights on its advantages and which projects are best suited for tubes over traditional trays and pouches.
Benefits of Using Tube Packaging
- Time & Cost Avoidance
- No custom tooling
- Off-the-shelf design
- Does not require heat sealing
- Minimal Footprint
- Clients see around a 50-75% footprint reduction when they switch from a pouch or a tray to a tube
- Saves shelf and operating room space
- Reduces sterilization costs
- Ease of transportation
- High Visibility & Traceability
- Clear tubes allow the end user to easily identify what is inside
- Ease of Use
- Intuitive design
- Aseptic Transfer
- Easy as twisting open a bottle of soda
- Aesthetics
- Appealing design
- High-quality & durability
- Single and dual barrier options
- Making it an option for a variety of projects
Design Inputs for Tube Packaging
Tube packaging is specifically designed with the end user, patient safety, sterile barrier integrity, and sustainability in mind. This evolution in packaging design was developed due to the demand from a spine surgeon who was looking for a better way to aseptically transfer screws in a sterile environment.
Tube packaging is hermetically sealed and allows for controlled aseptic transfer. They are specifically designed so they can’t be over-torqued to failure. These design specifications are all a product of listening to the voice of the intended user and understanding the importance of keeping the sterile barrier intact.
What are the alternatives to tube packaging?
The primary alternatives for tube packaging are trays and pouches. Medical device manufacturers who will most likely be considering tube packaging are typically designing devices such as cranial drills, orthopedic screws, nails, etc. These kinds of devices, which are often smaller and sharp, or long and heavy, might experience failures during testing if they’re simply dropped into a pouch. These failures might force the manufacturer to add extra packaging to prevent the device from damaging the sterile barrier. The extra packaging and additional tests required to validate these traditional packaging designs could be a costly endeavor.

Sustainability
We’ve already talked about how tube packaging offers a smaller footprint, but there’s other advantages:
- No secondary protective packaging
- Eliminating material and waste
- Enables a single barrier configuration
- No multilayer films
- Unlike pouches that often require 3–4 different materials, tubes are a mono-material structure.
- No Tyvek or glue
- Fewer components, less complexity, and lower costs.
- Recyclability
- Tubes can be recycled, reducing waste
The result? Lower transportation costs, more shelf space, and reduced environmental impact.
Key Takeaways
Tube packaging is a valuable option for any medical device manufacturer looking for a sleek, Gamma sterilized, easy to use packaging design for their sterile packaging strategy. The key benefits include minimal footprint, robust, repeatable, and reliable seal, and intuitive aseptic transfer. Tube packaging is an example of the kinds of solutions that can come from listening to the concerns of industry professionals and identifying possible areas of improvement.