We’re Your Packaging Partner.
PCL provides expertise in medical device packaging solutions.
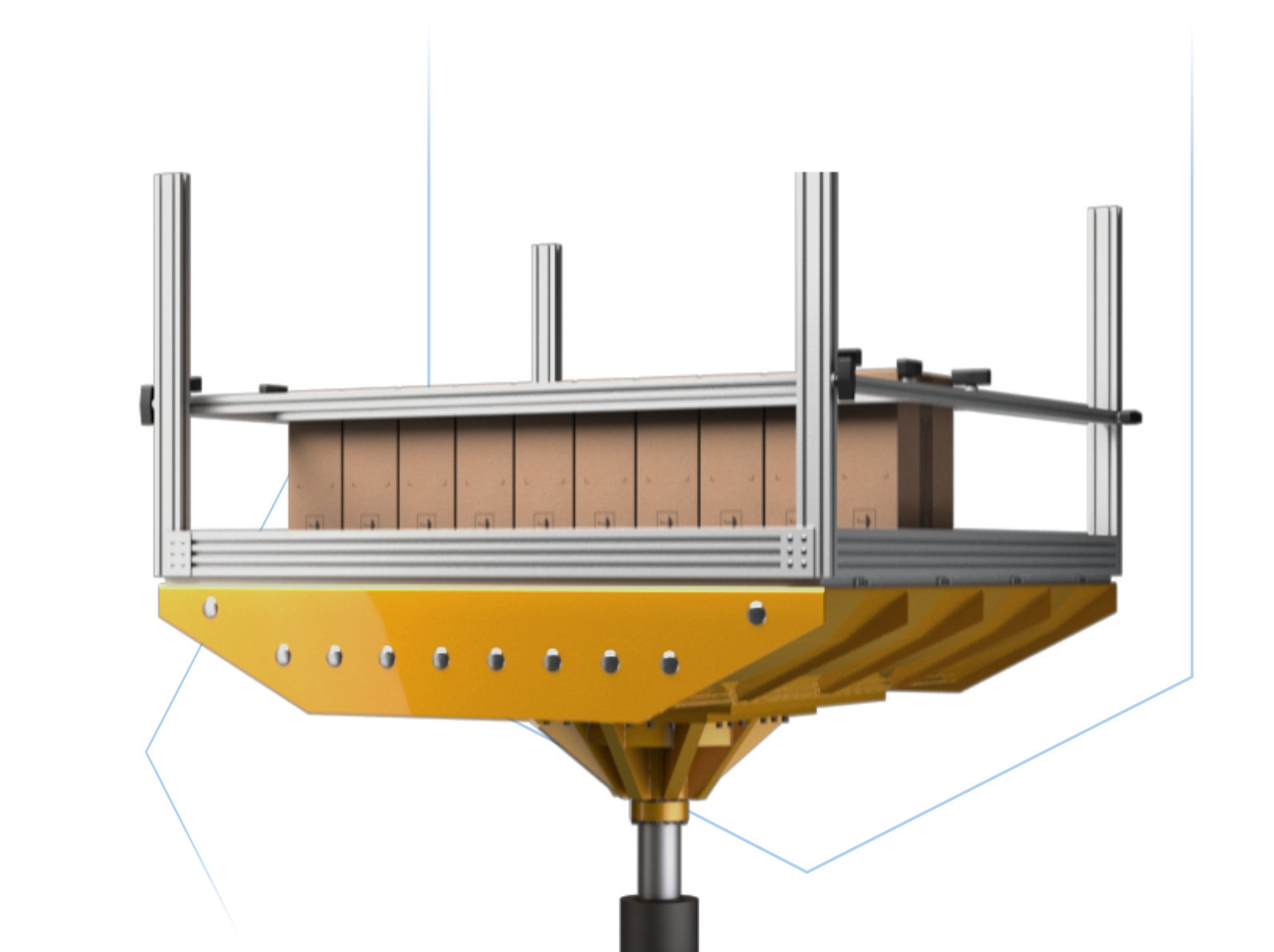
We are experts in sterile packaging
-
Package Testing
-
Contract Packaging
-
Packaging Engineering

Package Testing
Our ISO 17025 accredited package testing lab offers the full suite of ISO 11607 sterile package testing for medical devices.

Contract Packaging
PCM is ISO 13485-2016 certified and FDA registered. We specialize in low- to mid-volume contract packaging services.

Packaging Engineering
With PCL, you can take an a la carte approach that best fits the needs of your project – from custom packaging engineering to usability evaluations.
Our Accreditations
We partner with medical device manufacturers, development firms, and start-ups to deliver excellence in packaging.
-
ISO 17025-Accredited
-
ISTA Certified Testing Laboratory
-
ISO 13485 Certified
-
A2LA
The PCL Difference
In-house medical device packaging engineers
Our in-house team will serve as an extension of your team to strategize and innovate throughout the product life cycle.
Shorter lead times
We know how important package testing is to the product development life cycle because we live it every day. We’ve built our lab with that same sense of urgency and have systems in place to make sure we get it right the first time.
Medical device packaging expertise
Our entire business is focused on medical device packaging and our people are experts in their fields. We have our finger on the pulse of industry trends and regulatory inspections.
Quarterly Performance Targets
24hr
Dock to Start
95%
On-Time Delivery
48hr
Report Turnaround

Customer service is our version of patient care
When you partner with Packaging Compliance Labs, you’ll get honest insight, quick response and a collaborative team that is committed to delivering a customized solution that exceeds expectations. Learn more about how we can help you succeed.

Resources
Please feel free to use and share our resources about industry trends, standards and success stories.
View All Resources